Exclusive Insights from Cheryl K. Olson Sc.D. “It’s remarkable how little we know about how people stop smoking in everyday […] Read More

3 min. read


3 min. read
Exclusive Insights from Cheryl K. Olson Sc.D. “It’s remarkable how little we know about how people stop smoking in everyday […] Read More

2 min. read
Non-Targeted Chemical Analysis: Nontargeted chemical analysis is a powerful technique to simplify and understand the vastness of molecular complexity that […] Read More

6 min. read
We gave a presentation on the latest regulatory frameworks for cannabis products at the Keller and Heckman Symposium. This is […] Read More

2 min. read
Exclusive Insights from Cheryl K. Olson Sc.D. Where are we with ESG? Environmental, social and governance issues, that is. In […] Read More

4 min. read
By majority (10-6) opinion the United States Court of Appeal for the Fifth Circuit has determined that the Premarket Tobacco […] Read More

8 min. read
Enhancing Tobacco Harm Reduction through Holistic Approaches By Cheryl K. Olson “I firmly believe a lot of us, people like […] Read More

1 min. read
Understanding the Transformation: Unpacking International Tobacco Legislation, Upcoming TPD Changes, FDA CTP’s Role, and the Evolving Global Regulatory Scenario Read More

13 min. read
Hi, I’m Ian Fearon I’m the Director of WhatIF? Consulting Limited and Chief Scientific Officer with McKinney Regulatory Science Advisors. […] Read More
6 min. read
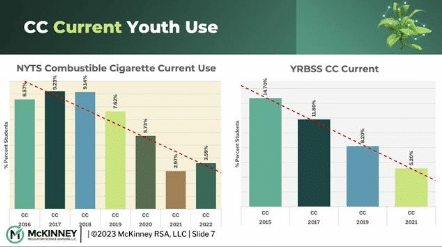
Hello, I am Mark Crosswhite, and I’m happy to be presenting data that were predominantly sourced from the youth risk […] Read More
"*" indicates required fields